Inteldevconference
Intel HPC Software WorkshopAn Introduction To Monoclonal Antibodies
Tuesday , 24, August 2021 Business and Management Comments Off on An Introduction To Monoclonal AntibodiesMonoclonal antibodies are a type of therapy that has been studied to treat COVID-19. These antibodies are often made by identifying pathogen-specific B cells in patients who have recently recovered from an infection or by immunizing genetically modified mice to have humanized immune systems and collecting strong antibodies from them.
Once B cells were identified, the immunoglobulin heavy and light chain genes were restored. These genes are then expressed to produce monoclonal antibodies. If you want to know more about monoclonal antibodies, you can also visit https://www.bosterbio.com/featured-products.
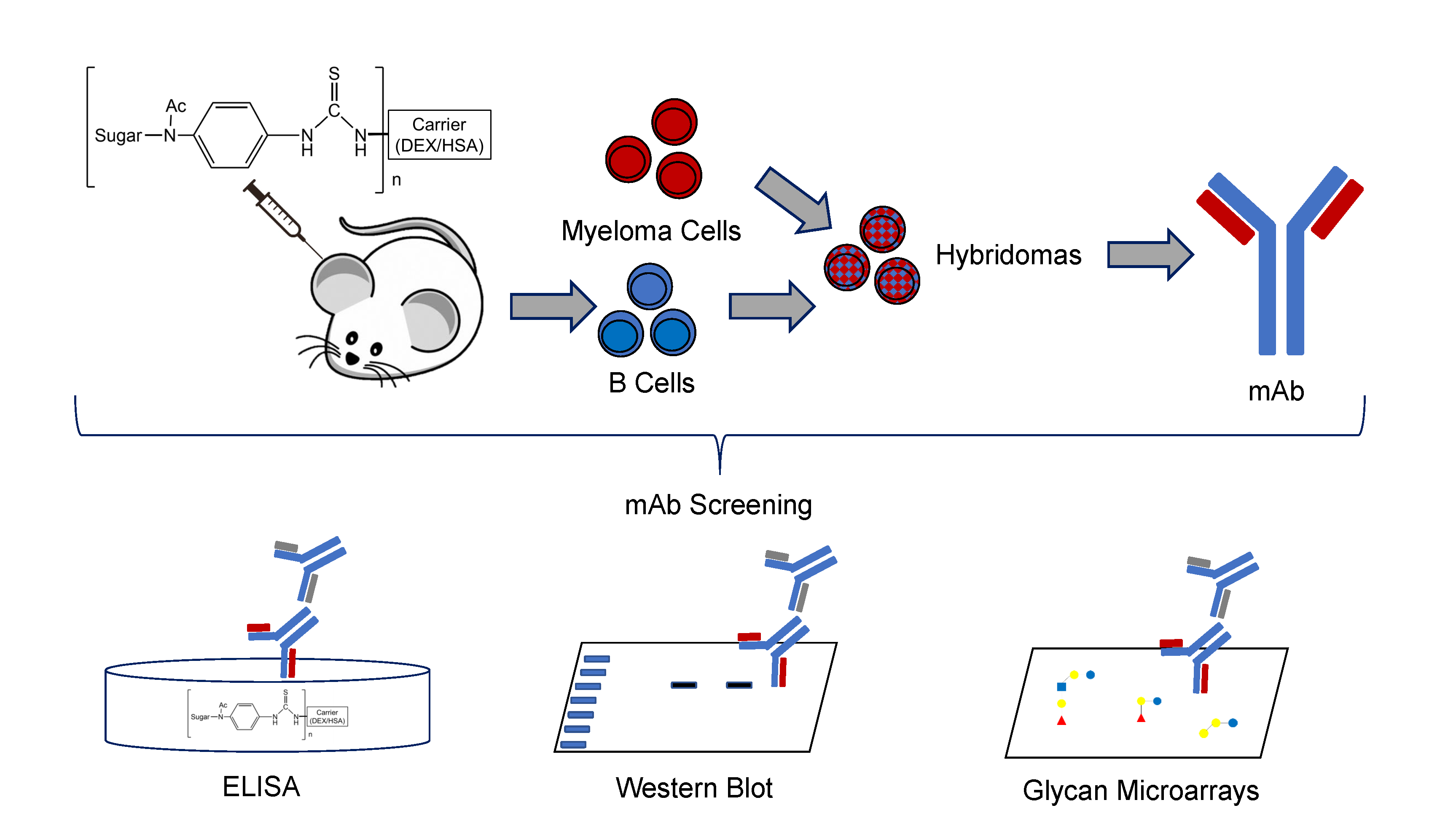
Image Source: Google
Monoclonal antibodies have specific activity against a predetermined target; therefore, they differ from convalescent plasma, which consists of polyclonal antibodies in the serum of patients recovering from infection.
Monoclonal antibodies have been developed to treat and prevent other viral infections such as HIV, influenza, RSV, MERS-CoV, Ebola, and Zika.
Of these, only monoclonal antibodies against RSV and Ebola have been shown to be effective in human studies (the first to be FDA-approved). Several products targeting the other viruses listed above are currently being investigated in clinical trials.
Most of the direct antiviral monoclonal antibody products developed for SARS-CoV-2 target the spike protein that the virus uses to enter host cells, thereby blocking the attachment and entry of the virus into human cells.